2004 Drosophila Image Award
Winner
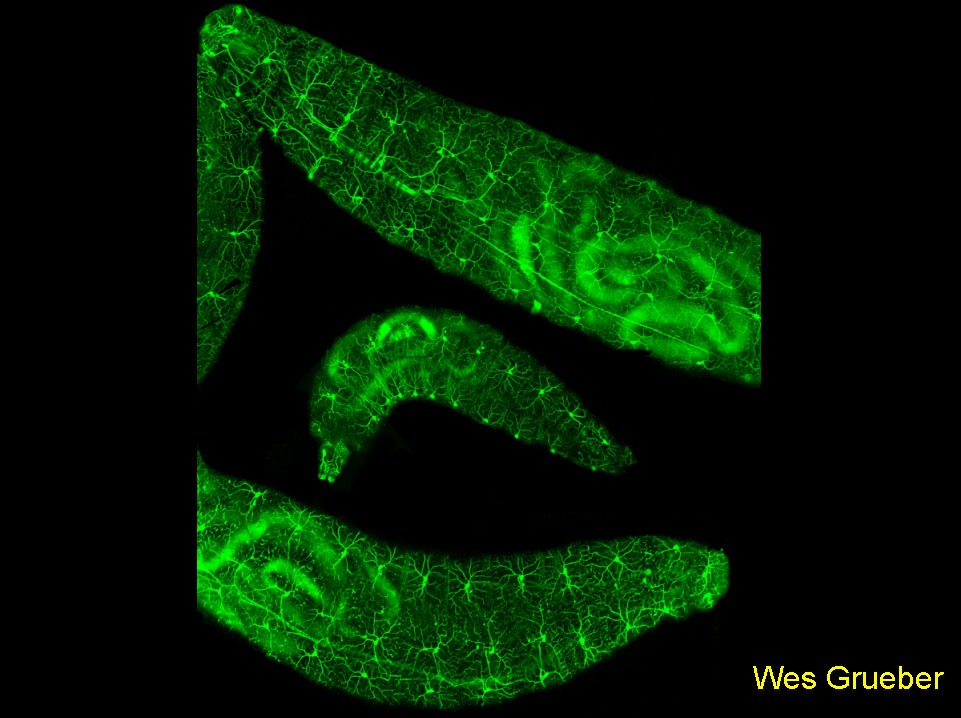
The image shows a confocal montage of Drosophila larvae expressing EGFP under the control of a 1kb genomic region 5' of the pickpocket gene. Labeling is observed in a segmentally repeated subset of dendritic arborization (da) sensory neurons. The dendrites of these neurons provide a complete and non-redundant tiling of the epidermis (axons can be seen projecting to the CNS). Ablation experiments suggest that homotypic dendrites repel each other where they meet.
Grueber WB, Ye B, Moore AM, Jan LY, Jan YN. 2003. Dendrites of distinct classes of Drosophila sensory neurons show different capacities for homotypic repulsion. Curr Biol 13:618-626.
Runner-up
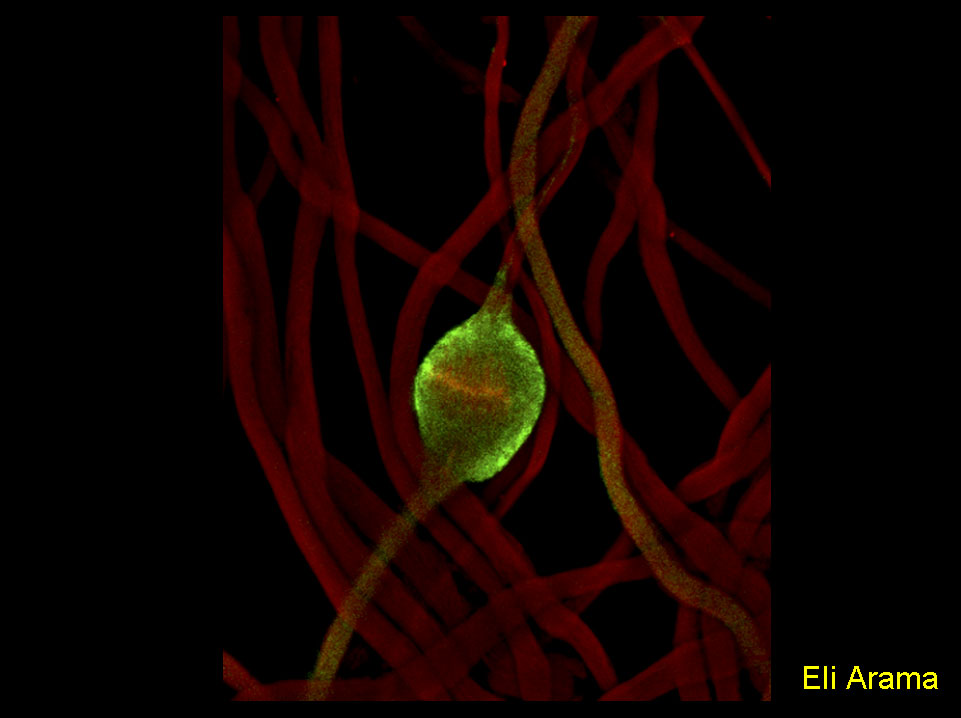
A new, non-cell killing role for the apoptotic machinery.
Maturing spermatids utilize components of the apoptotic machinery to remove unneeded cytoplasmic contents during the process of spermatid individualization.
The figure shows Drosophila spermatids in the process of removing their bulk cytoplasm. The expelled cytoplasm is collected into a ‘waste bag’ (marked by the red filament staining of the individualization complex) by an apoptosis-like process that requires caspase activity (activated drICE, green).
Arama E, Agapite J, and Steller H. Caspase activity and a specific cytochrome C are required for sperm differentiation inDrosophila. Dev Cell. 2003 May;4(5):687-97.
Runner-Up
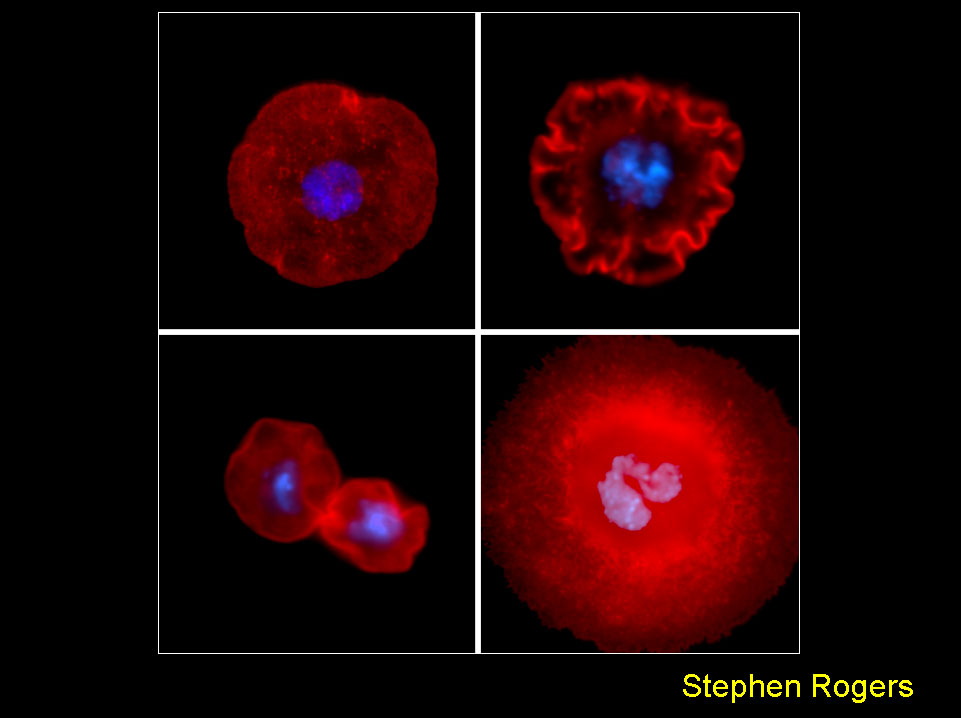
Drosophila S2 cells offer a powerful tool to study in vivo dynamics and organization of the actin cytoskeleton. When plated on the lectin, concanavalin A, S2 cells attach and spread on the substrate to form a circumferential actin-based lamellae. The susceptibility of these cells to gene inhibition using RNAi makes them a very tractable system to dissect the molecular machinery involved in lamellipod formation. The figure shows a control S2 (upper left) in comparison with cells depleted of capping protein beta (upper right), cofilin (lower left), and Rho1 (lower right). The hyper-ruffled morphology produced by capping protein RNAi is consistent with its role in terminating actin filament elongation - in the absence of the protein, actin filaments polymerization pushes on the membrane in an unregulated manner. Depletion of cofilin, a factor required for actin filament turnover, produces cells that are unable to spread due to abnormal accumulations of f-actin at their cortex. RNAi-inhibition of Rho1 does not inhibit lamellae formation, but results in a cytokinesis failure that produces giant, multinucleate cells.
Rogers et al., J Cell Biol. 162: 1079-1088 (2003).
Finalist
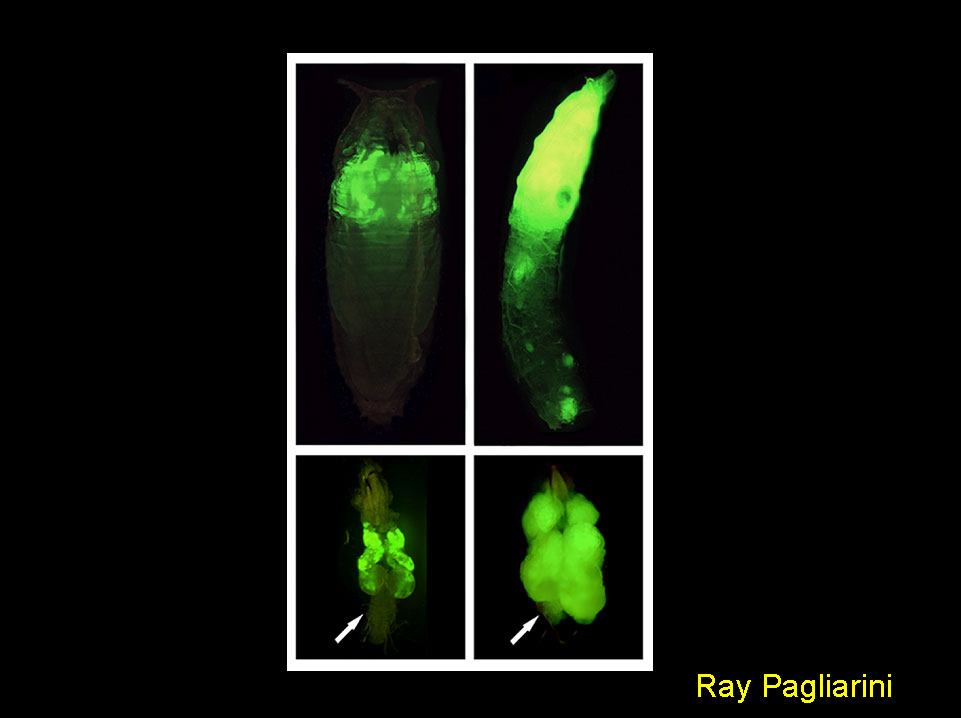
Metastatic behavior in Drosophila.
GFP-labeled tumors made specifically in the eye through expression of oncogenic Ras do not form secondary growths (left top panel) and do not invade adjacent tissues such as the larval central nervous system (arrow, left bottom panel). Inactivation of cell polarity genes such as scribble causes the appearance of secondary growths (right top panel) as well as the invasion of cells into the central nervous system (arrow, right bottom panel). As neither oncogenic Ras nor scribble inactivation can cause this phenotype on their own, both alterations cooperate to promote metastatic behavior. This genetic model for metastasis allows for the systematic discovery of genes and cellular processes that may play a role in human malignant cancers.
R.A. Pagliarini and T. Xu. A Genetic Screen in Drosophila for Metastatic Behavior.
Science. 2003 302:1227-1231
Finalist
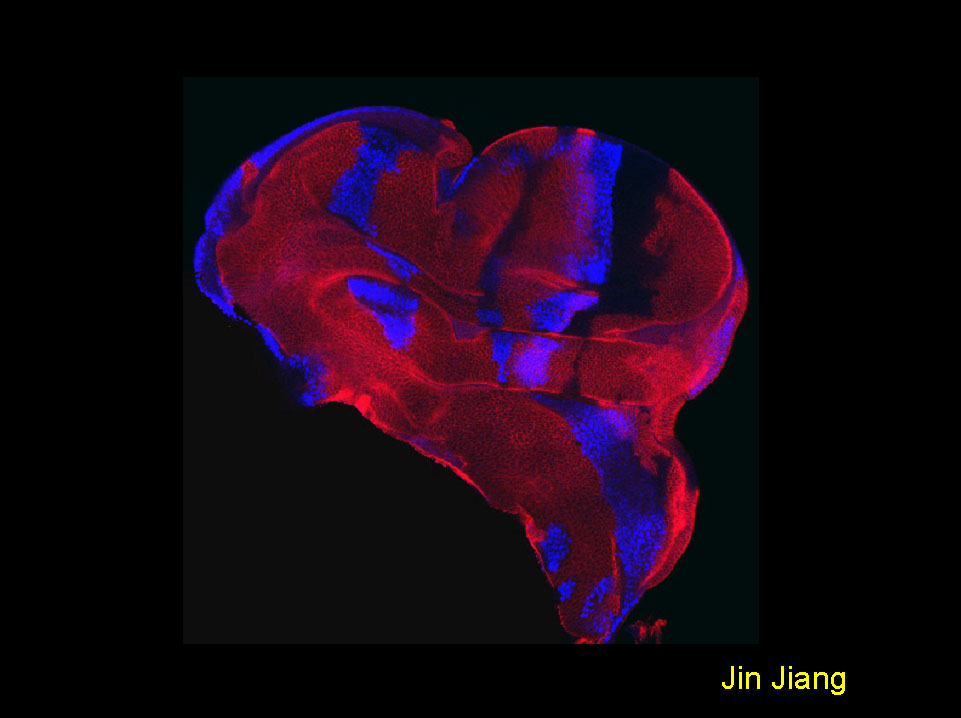
Smoothened transduces the Hedgehog signal by physically interacting with Costal2/Fused complex through its carboxy-terminal tail
A late third instar wing disc carrying clones of cells expressing a membrane tethered Smoothened (Smo) carboxy-terminal tail (Myr-SmoCT) under the control of act>CD2>Gal4. The disc was stained to show the expression of Hh target gene dpp-lacZ (blue) and a marker gene CD2 (red). Cells expressing Myr-SmoCT do not express CD2, hence are recognized by the lack of CD2 expression. Expressing Myr-SmoCT in anteriorly-situated cells elicits Hedgehog pathway activation, as indicated by the ectopic expression of dpp-lacZ.
Jianhang Jia, Chao Tong, and Jin Jiang. 2003. Smoothened transduces Hedgehog signal by physically interacting with Costal2/Fused complex through its carboxyl-terminal tail. Genes & Development 17, 2514-2519.
Finalist
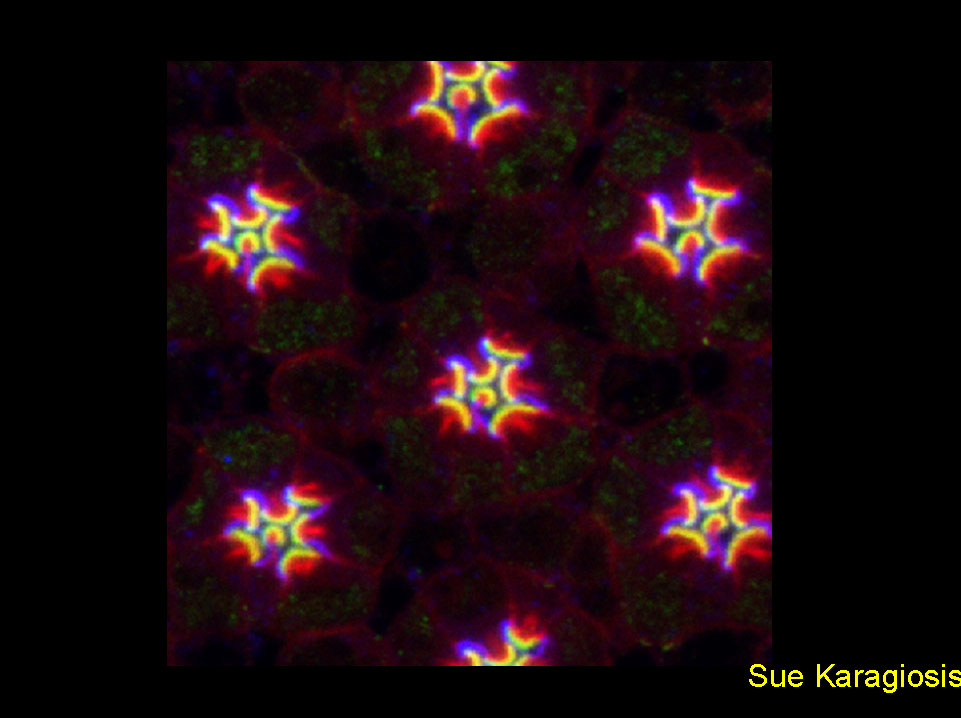
Crumbs and Moesin define Drosophila photoreceptor apical domains
Confocal micrograph of a 57% pupal development retina shows immunostaining for Moesin (green) and Crumbs (blue). The actin cytoskeleton was stained with rhodamine-conjugated phalloidin (red), highlighting the dense bundles of the rhabdomere terminal web. Adult Drosophila photoreceptor apical plasma membranes are organized into complementary domains: the photosensory rhabdomere and an adjacent supporting membrane, the stalk. Definitive rhabdomere and stalk primordia are established within the photoreceptor apical membrane at approximately 50% of pupal development. Before this stage, Moesin and Crumbs extend fully across the apical surface. Concomitant with morphological differentiation, Moesin and Crumbs resolve to their respective adult domains.
Karagiosis SA, Ready DF. Crumbs and Moesin define Drosophila photoreceptor apical domains NAT CELL BIOL 5 (6): 504-504 JUN 2003
Finalist
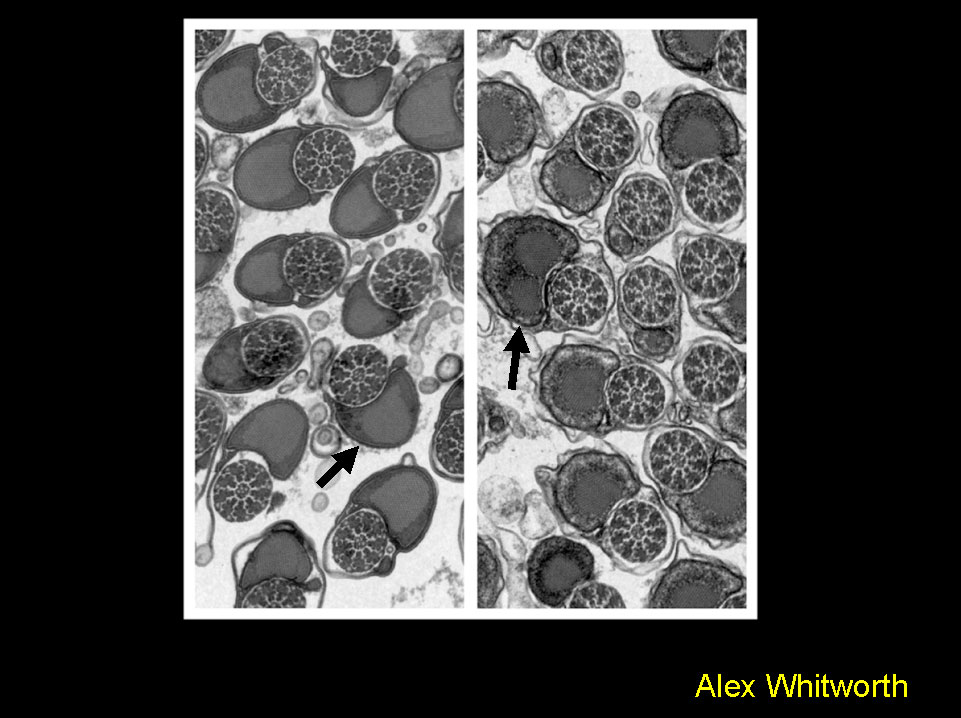
Phenotypic analysis of the Drosophila homolog of Parkin - a gene linked to Parkinson’s disease.
Electron micrograph showing a cross section through tails of mature spermatids from wild type (left) and parkin mutants (right). Loss of parkin function causes mitochondrial dysfunction, shown here by the disruption of the mitochondrial derivative, the Nebernkern (arrows).
This evidence links a genetic mutation to mitochondrial dysfunction, previously biochemically implicated in the etiology of Parkinson’s disease
Greene et al., 2003. PNAS, 100: 4078-4083.
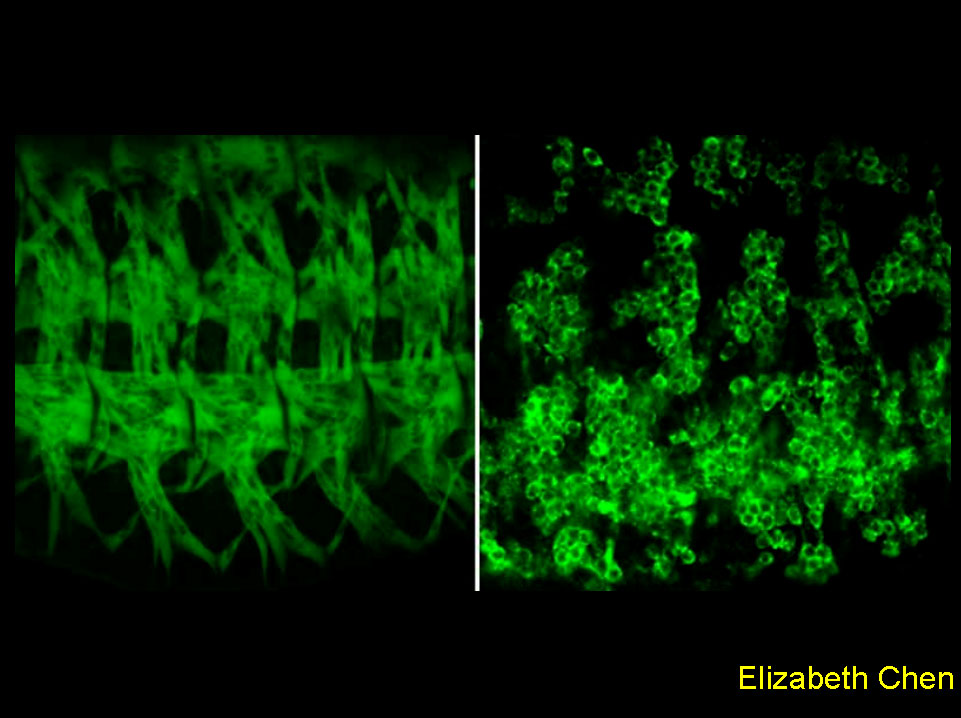
Finalist
This image demonstrates the requirement of a novel guanine nucleotide exchange factor, Loner, in myoblast fusion duringDrosophila muscle development. The left panel shows the somatic musculature of a wild-type embryo, visualized by a muscle-specific GFP reporter. Each muscle fiber is a product of fusion between a certain number of myoblasts. The right panel shows differentiated muscle cells in a loner mutant embryo. The presence of a massive amount of mononucleated, unfused myoblasts indicates a defect in myoblast fusion.
Chen et al., (2003). Cell 114: 751-762
Finalist
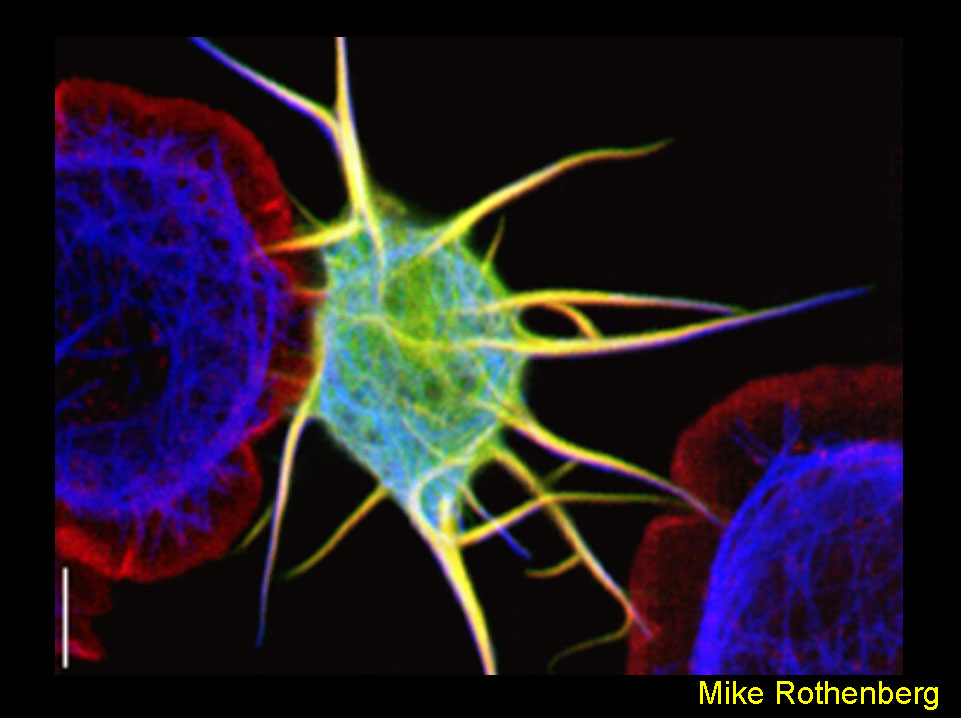
Remodeling of Cell Shape by Overexpression of Dpod1
Overexpression of Dpod1GFP (green) in transfected DrosophilaS2 cells causes the outgrowth of neurite-like processes (center cell). Untransfected cells (left and right cells, non-green) are discoid with no processes. Cells were transfected, plated onto concanavalinA-coated cover slips, and stained for actin (red) and microtubules (blue). A Leica TCS-SP2 Confocal microscope was used. Bar=10µM. The author thanks Wes Grueber for assistance with image processing.
Rothenberg ME, Rogers SL, Vale RD, Jan LY, and Jan YN.Drosophila Pod-1 Crosslinks Both Actin and Microtubules and Controls the Targeting of Axons. Neuron (2003) 39:779-791.
Finalist
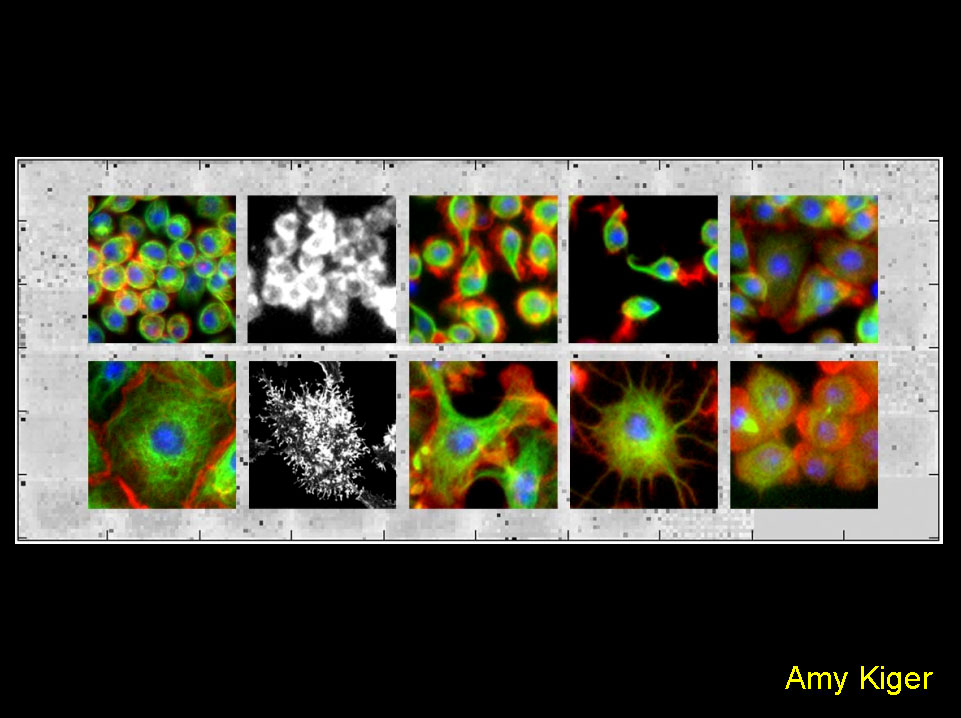
Systematic RNAi screens in Drosophila cells identify known and novel gene functions required for specific cell-biological processes.
Foreground: Systematic RNAi screens in cultured Drosophila cells identified gene functions associated with distinct morphological phenotypes. Automated fluorescence microscopy was used to detect organization of filamentous-actin (red), microtubules (alpha-tubulin, green) and DNA (blue), allowing unprecedented genetic dissection of cellular morphogenesis. Specific defects included those in cell adhesion, lamellipodia formation, cell cycle progression and cytokinesis.
Kiger, A. A., B. Baum, S. Jones, M. Jones, A. Coulson, C. Echeverri, N. Perrimon. 2003. A functional genomic analysis of cell morphology using RNA-interference. J. Biol., 2: 27.