2026 Drosophila Image Award
Winner - Video
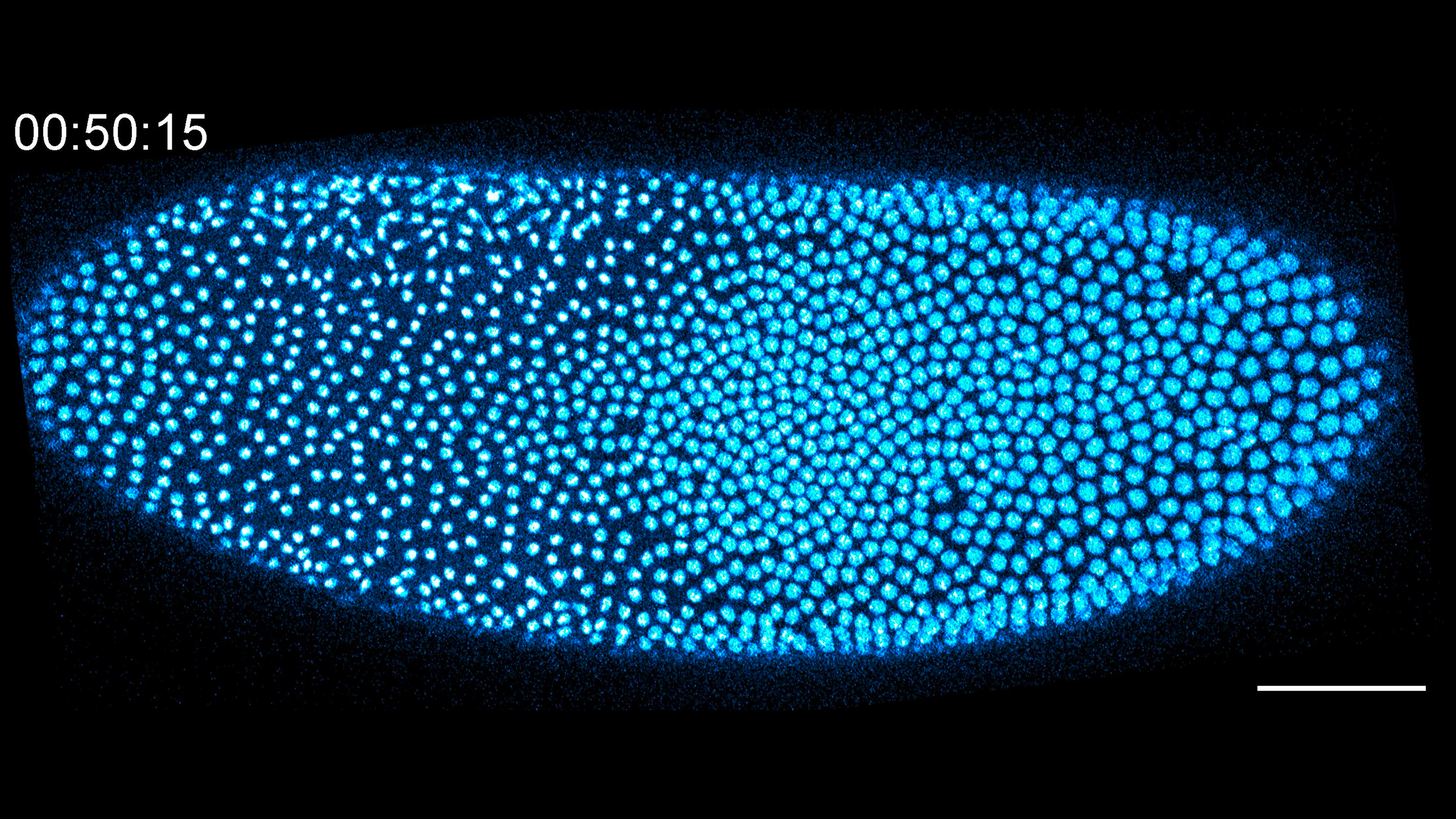
Making the germline
This video shows the coordination between the germline-determinant germ granules and the F-actin cortex during formation of the primordial germ cells. We find that germ granules prime the F-actin cortex in their immediate vicinity, enriching F-actin at the posterior pole in preparation for arrival of the syncytial nuclei at nuclear cycle 9. As the nuclei arrive, they collect the germ granules, triggering an F-actin transition to form a cohesive dome structure that is propelled outward by buckling forces. As the buds emerge, the nuclei divide before fully pinching off from the syncytium. The video is a Z-projection of the posterior pole; F-actin is shown in magenta while germ granules are shown in green. Scale bar: 10 μm
Video credit: Marcus Kilwein
Marcus D. Kilwein, Pearson Miller, Kwan Yin Lee, Miriam Osterfield, Alex Mogilner, Stanislav Y. Shvartsman, and Elizabeth R. Gavis.
Formation of Drosophila germ cells requires spatial patterning of phospholipids
Current Biology, Volume 35, Issue 7, 1612 - 1621.e3 10.1016/j.cub.2025.01.071
Winner - Still Image
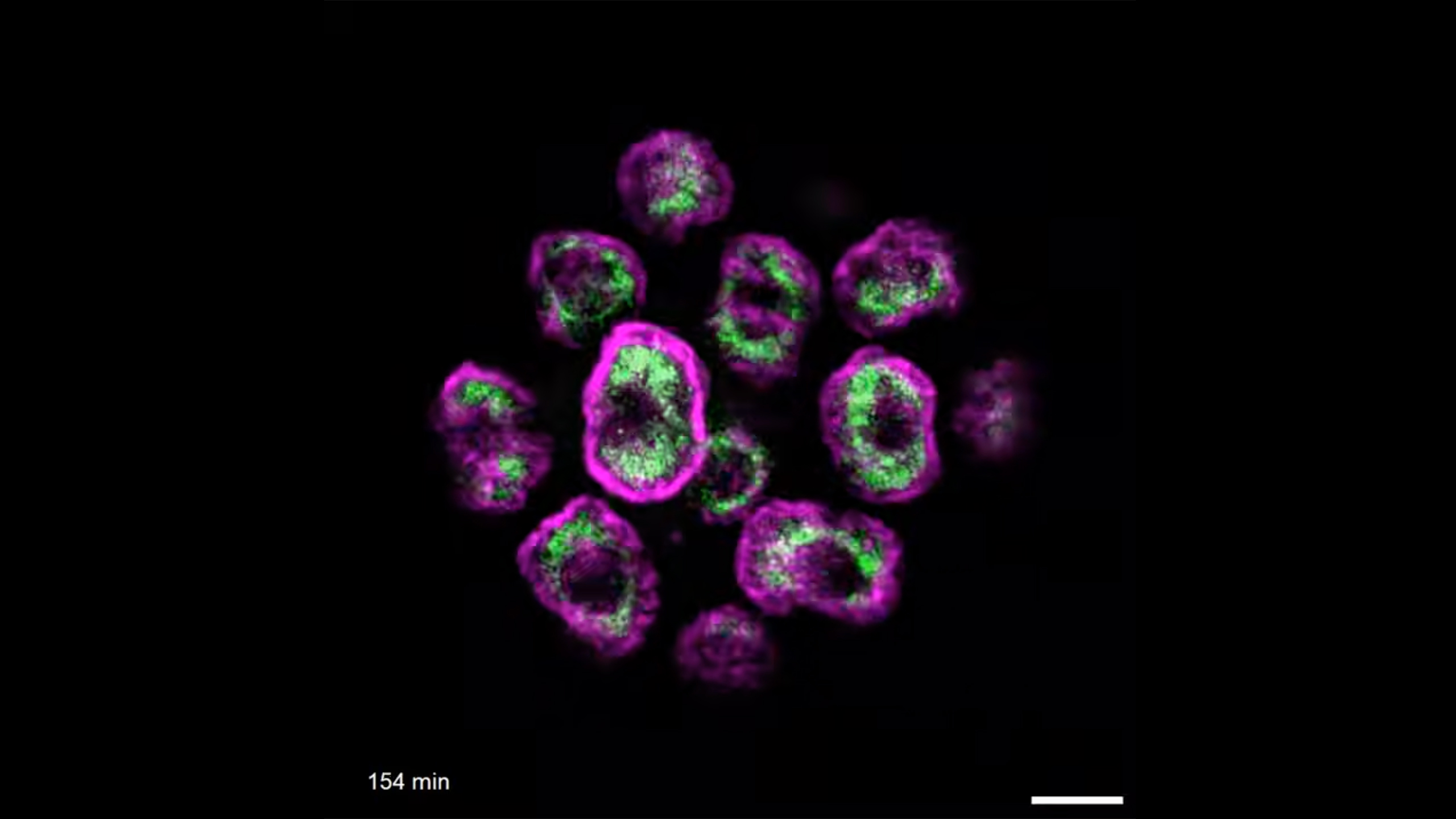
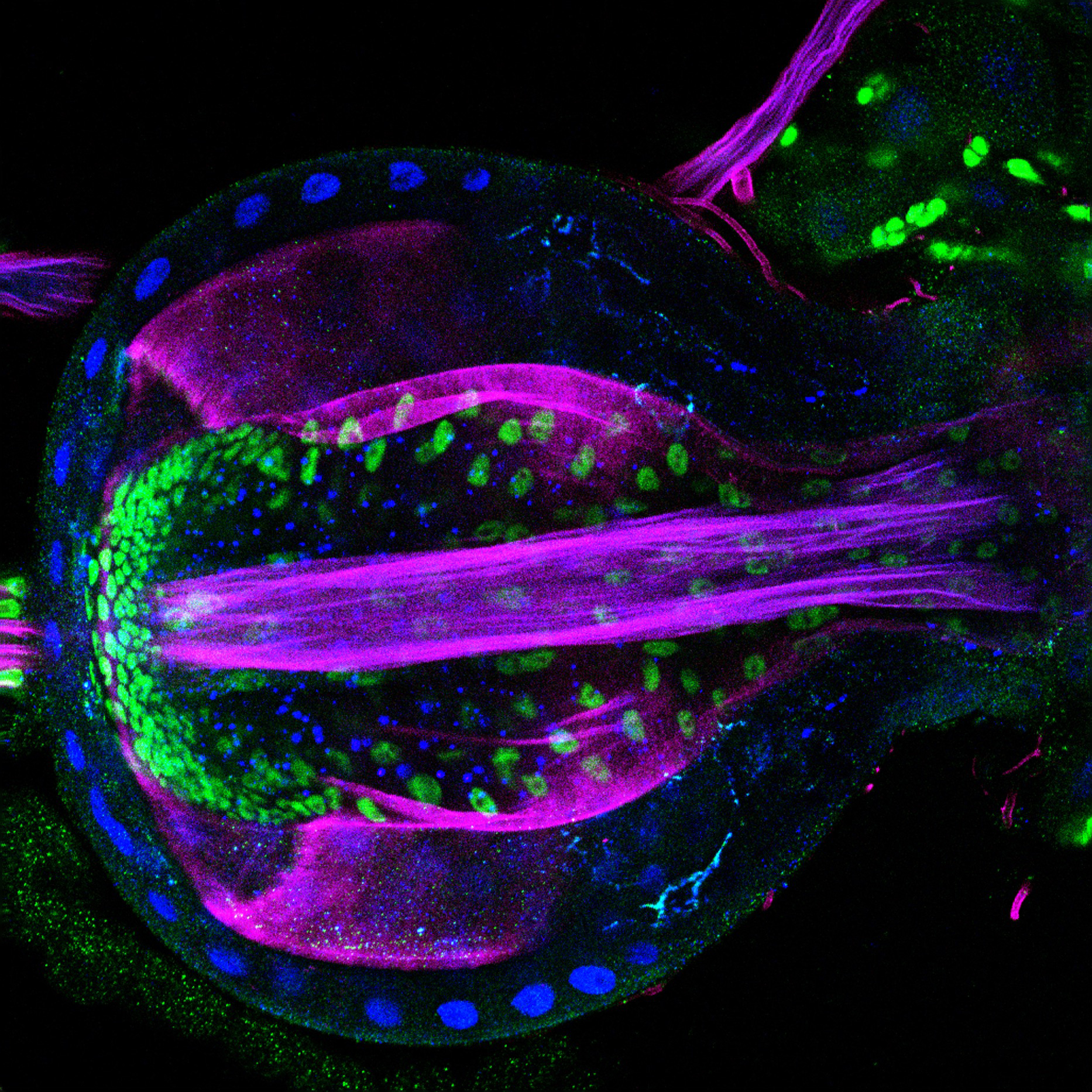
Dynamic nuclear adaptation by immune cells during in vivo confinement
Cells navigating in complex 3D environments often encounter narrow spaces that physically challenge their migration. Here, we harnessed the Drosophila pupal wing to explore how immune cells adapt to confinement in vivo. Using live confocal microscopy, this image captures immune cells (magenta, srp driven mCherry) migrating within narrow wing vessels (epithelial GFP-Moesin, cyan), where vessel confinement triggers nuclear deformation (inset; white, nuclear RFP). Strikingly, we find that vessel-bound immune cells adapt their nuclear lamina composition to enhance nuclear deformability, curb damage (e.g. nuclear rupture), and thereby optimise motility in vivo.
Image Credit: Tua Karling
Tua Karling and Helen Weavers
Immune cells adapt to confined environments in vivo to optimise nuclear plasticity for migration.
EMBO Reports 26, 1238-1268 (2025)
https://link.springer.com/article/10.1038/s44319-025-00381-0
1st Runner Up - Video
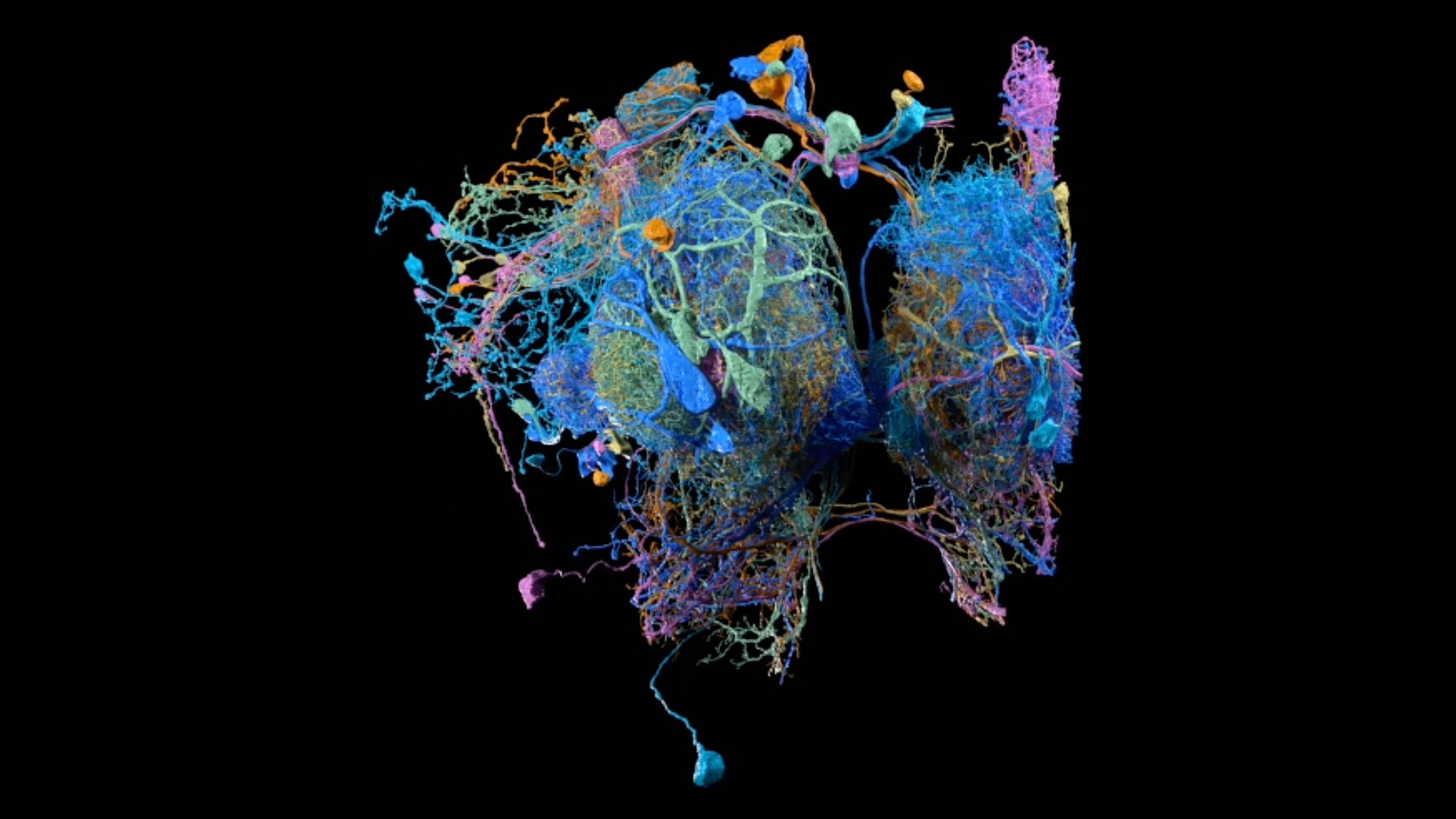
A Striking Ladder-Like Architecture of the Endoplasmic Reticulum in Drosophila Neurons
The endoplasmic reticulum (ER) is a master organizer of intracellular communication. It is a vast organelle that extends as a continuous structure from the perinuclear region to the cell periphery, yet its architecture in neurites has remained elusive due to the narrow caliber of its tubules and their extreme dynamism. Inspection of volume electron microscopy data spanning the fly brain reveals that the neuronal ER possesses a striking and unexpected morphology. This video zooms into the neurite of an MBON1 neuron in the Drosophila brain and shows 3D renderings of the endoplasmic reticulum (ER, green), plasma membrane (blue), mitochondria (pink), microtubules (tan), and ER–plasma membrane contact sites (magenta), segmented from FIB-SEM datasets.
Video credit: Philip Hubbard
Volume EM dataset: Harald Hess Lab and the FlyEM project team
Organelle segmentation: Aubrey V. Weigel, Grace Park, Alyson Petruncio, Stephan Saalfeld Lab and the CellMap project team
Benedetti L, Fan R, Weigel A, Moore AS, Houlihan PR, Kittisopikul MA, Park G, Petruncio A, Hubbard P, Pang S, Xu CS, Hess H, Saalfeld S, Rangaraju V, Clapham DE, De Camilli P, Ryan TA, Lippincott-Schwartz J.
Periodic ER-plasma membrane junctions support long-range Ca2+ signal integration in dendrites.
Cell. 2025 Jan 23;188(2):484-500.e22. 10.1016/j.cell.2024.11.029
1st Runner Up - Still Image
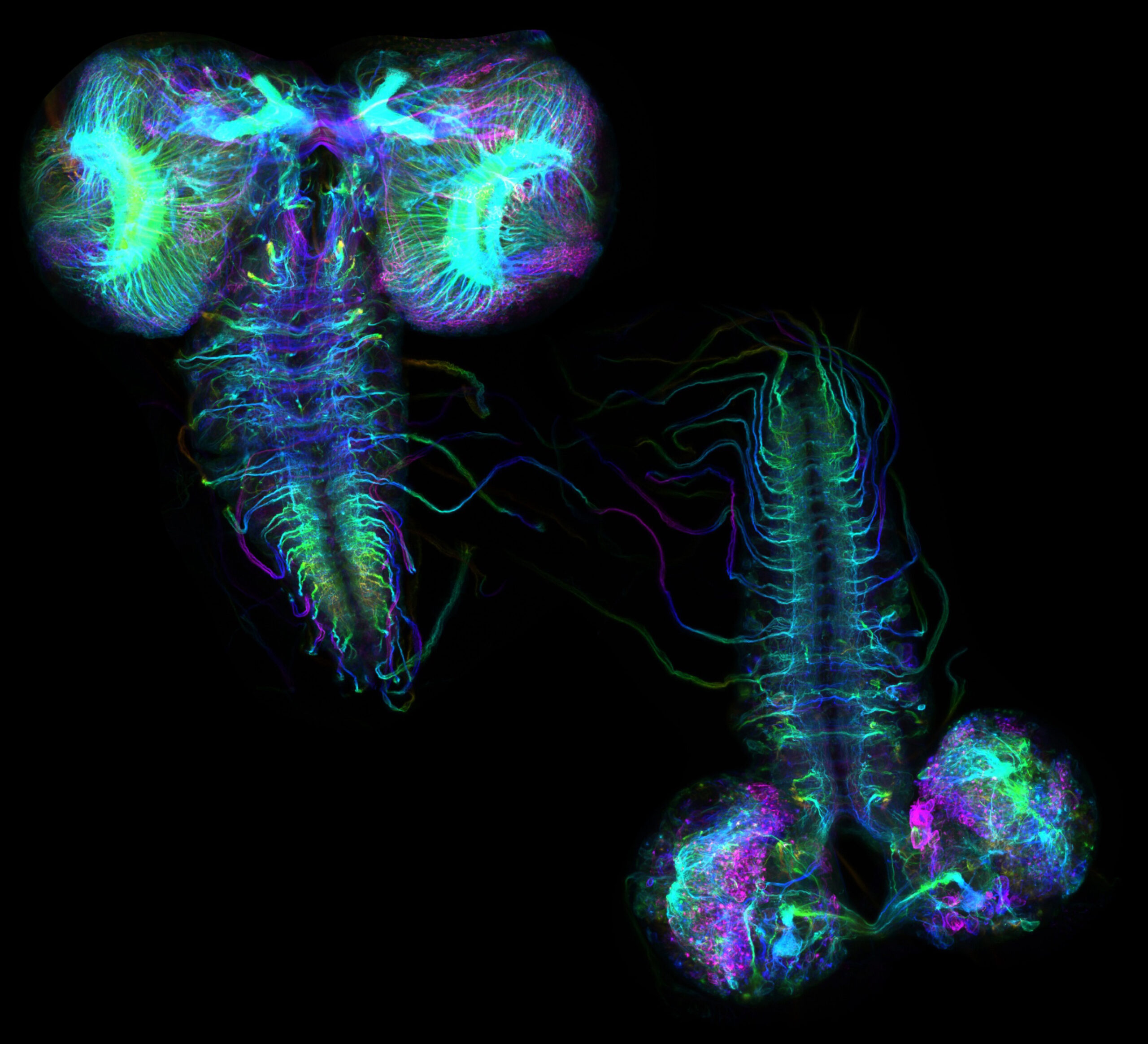
Kinesin-5 acts as a dynamic brake on axonal wiring in the Drosophila larval brain
Neuronal circuits form through a delicate balance between microtubule-driven growth and mechanisms that restrain it. Shown are spectrum color–coded hyperstacks assembled from z-stack images of Drosophila larval brains labeled with photoconvertible EOS-tagged tubulin, visualizing the 3D spatial organization of neuronal projections. Compared with control (left), neuron-specific knockdown of a mitotic motor, Klp61F (kinesin-5) (right), produces strikingly disorganized connectivity, consistent with kinesin-5 functioning as a “dynamic brake” that limits microtubule sliding to ensure precise axon growth and pathfinding.
Image credit: Wen Lu
Lu, W., Lee, B.S., Deng, H.X.Y., Lakonishok, M., Martin-Blanco, E., Gelfand, V.I.
'Mitotic' kinesin-5 is a dynamic brake for axonal growth in Drosophila.
Development (2025) 152(9): dev204424. doi: 10.1242/dev.204424
https://doi.org/10.1242/dev.204424
2nd Runner Up - Video
Shackleton (Shkl) mutant embryo undergoing asynchronous nuclear divisions
Shkl mutants have uneven nuclear to cytoplasmic (N/C) ratios, which result in asynchronous divisions along the A-P axis. This asymmetry was used to study the effect of the N/C ratio in regulating histone H3 variant composition on chromatin during zygotic genome activation. In this embryo, the posterior pole undergoes a partial extra division to even out the N/C ratio. The embryo expresses fluorescently tagged H3.3-Denrdra2.
Video credit: Anusha Bhatt
Anusha D. Bhatt, Madeleine G. Brown, Aurora B. Wackford, Yuki Shindo, and Amanda A. Amodeo
Local nuclear to cytoplasmic ratio regulates H3. 3 incorporation via cell cycle state during zygotic genome activation.
EMBO reports 26, no. 23: 5703.
https://link.springer.com/article/10.1038/s44319-025-00596-1
2nd Runner Up - Still Image
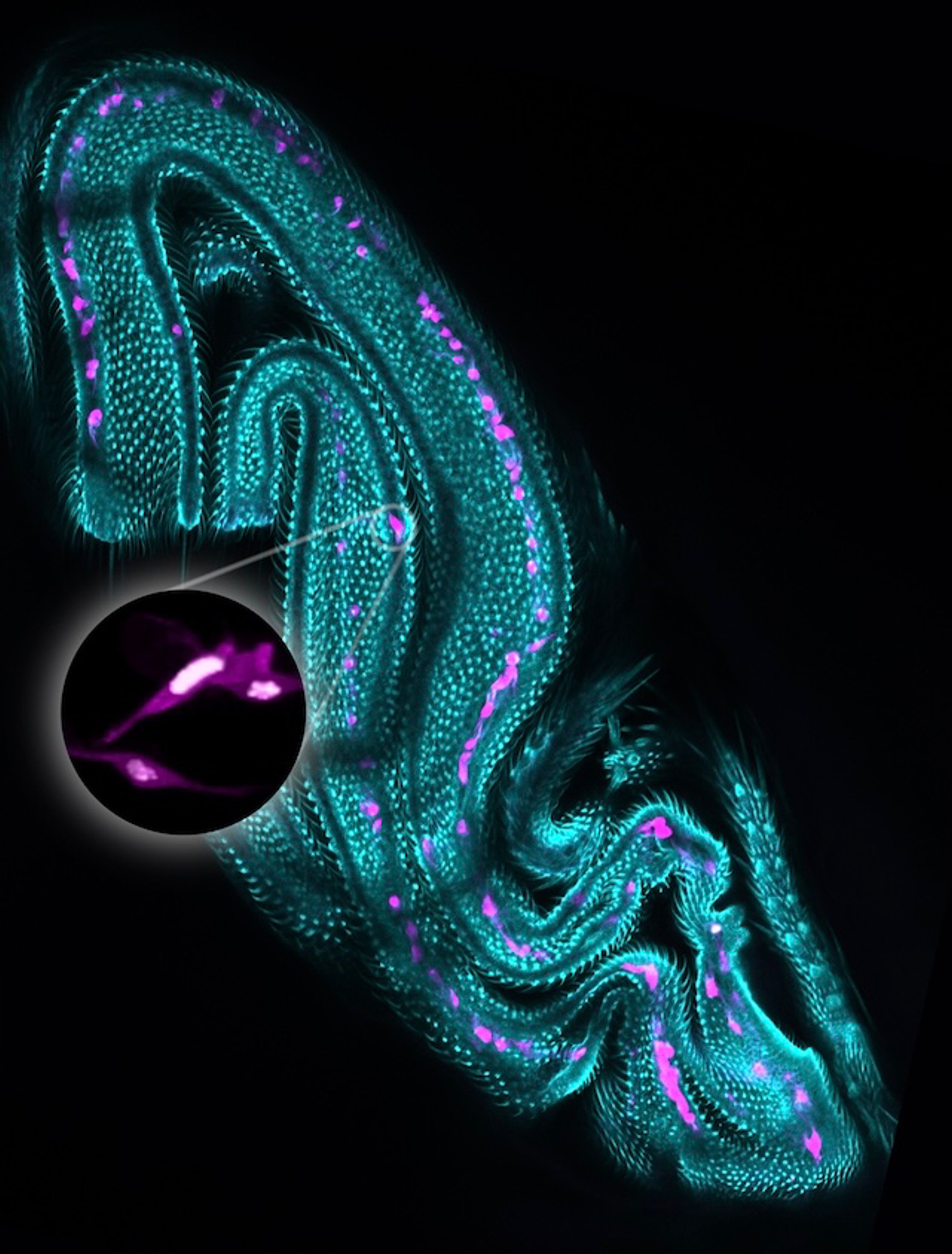
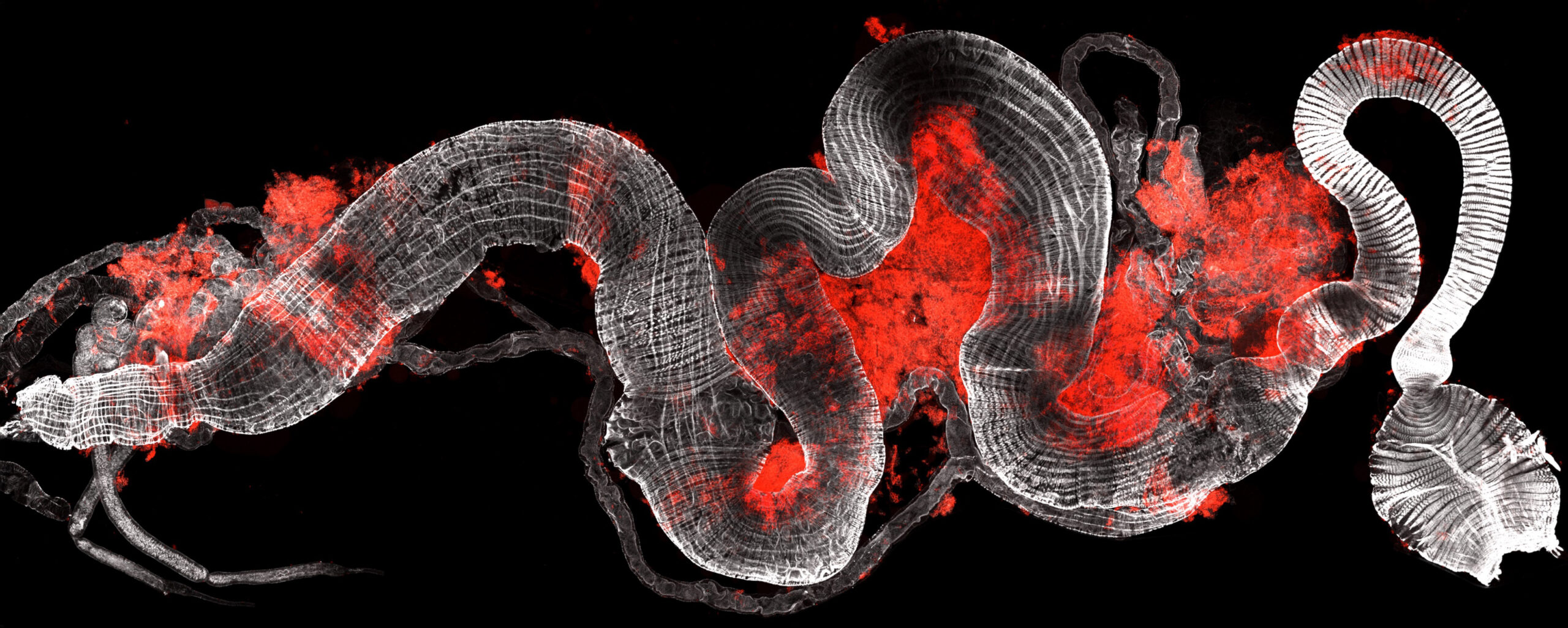
The aECM in the proventriculus of Drosophila larva
The Proventriculus is a complex bulb-shaped organ at the foregut-midgut junction that functions as a valve controlling food passage and produces the peritrophic matrix that lines the intestine. The organ is a composite of ectodermal and endodermal cells.
The image shows a confocal projection of a wild type proventriculus of a Larva 3 stained for Forkhead (in green) as a marker of ectodermal cells and for Defective proventriculus (in blue) as a marker of endodermal cells. CBP labelling of chitin (in magenta) reveals a halo in contact with the blue-endodermal cells, corresponding to chitin deposited in the peritrophic matrix, and a labelling tightly attached to the green-ectodermal cells, corresponding to the cuticle.
Image Credit: Marta Llimargas
Bertran-Mas J, De Giorgio E, Martín N, Llimargas M.
Distinct cellular and molecular mechanisms contribute to the specificity of the two Drosophila melanogaster chitin synthases in chitin deposition.
PLoS Genet. 2025 Sep 8;21(9):e1011847. doi: 10.1371/journal.pgen.1011847.
https://journals.plos.org/plosgenetics/article?id=10.1371/journal.pgen.1011847
Honorable Mention - Video
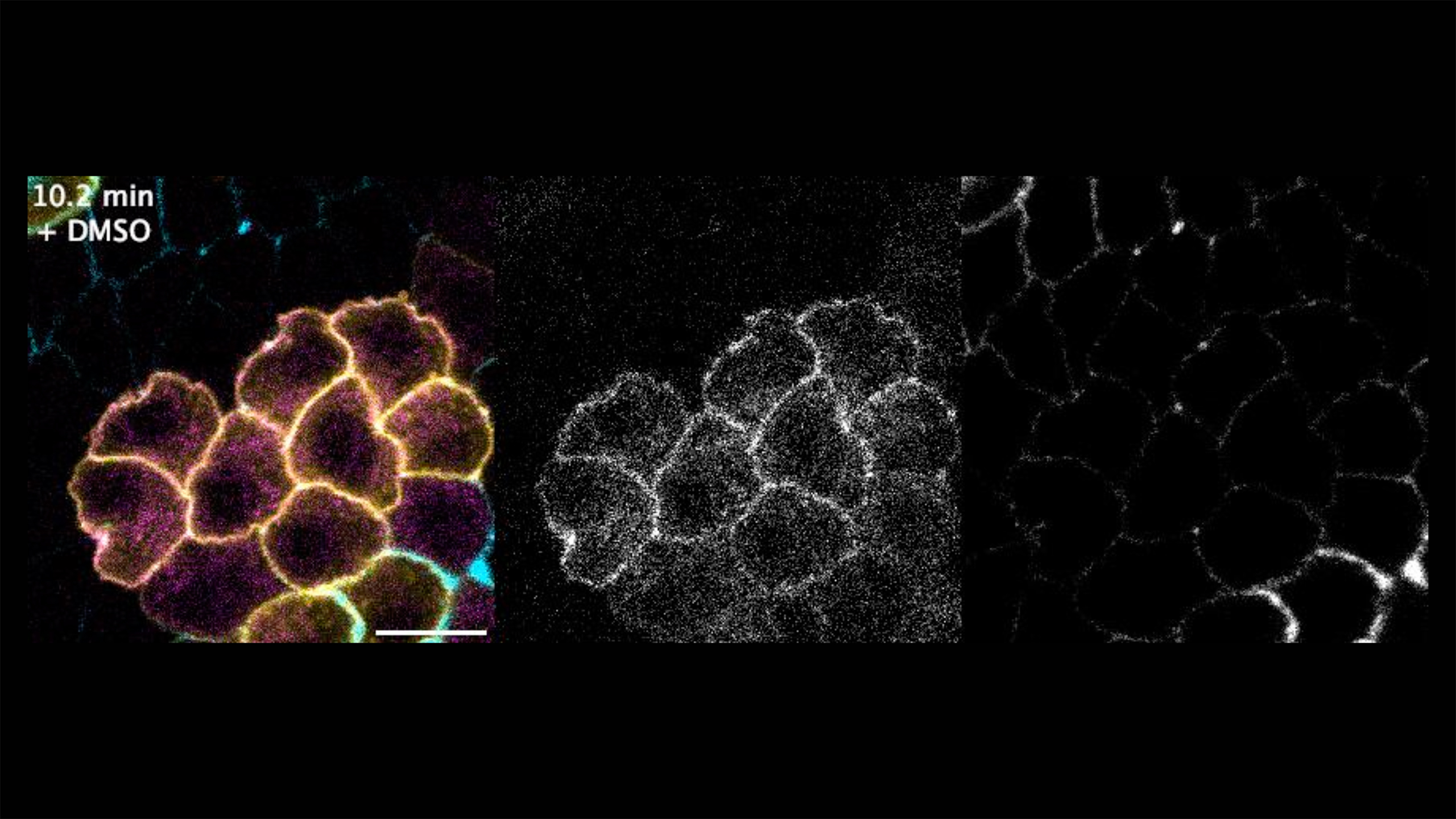
Rho signaling prevents intercellular gap opening in the follicle epithelium
Live imaging of a basolateral section of the follicle epithelium in a vitellogenic egg chamber. A patch of cells expresses the optoRhoGEF system (CIBN::pmGFP in yellow, CRY2-RhoGEF::mCherry in magenta, grey in middle panel). Fluorescently labelled dextran (cyan, grey in right panel) labels intercellular spaces. Exposure to blue light (t = 0 min) activates Rho signaling (note translocation of CRY2-RhoGEF::mCherry to the plasma membrane). Upon application of a hypersomotic shock (at t = 10 min), intercellular channels open at tricellular junctions in the wildtype epithelium, while no gaps open in the clone with active Rho signaling. This indicates that an increase of contractility is sufficient to prevent the opening of intercellular gaps.
Video credit: Thea Jacobs
Thea Jacobs, Jone Isasti Sanchez, Steven Reger and Stefan Luschnig
Rho/Rok-dependent regulation of actomyosin contractility at tricellular junctions restricts epithelial permeability in Drosophila.
Jacobs et al., 2025, Current Biology
https://doi.org/10.1016/j.cub.2025.01.043
Honorable Mention - Still Image
Imaging of of actin filament orientation and alignment in a living cellularizing embryo
Fluorescence intensity images of actin filaments at the bases of the cellularizing epithelium in the Drosophila embryo show that actin localizes in ring structures (left panels) but do not provide information on how actin filaments organize within the rings. We have engineered novel actin probes that now report actin filament orientation (middle panels) and alignment (right panels) per image pixel using fluorescence polarization microscopy. Filament orientation is represented as sticks, showing that highly-aligned actin filaments are oriented circumferentially along the ring contour. The engineered probes are genetically encoded and thus allow us to correlate actin filament organization in any living tissue, healthy or mutant, to the developmental process of interest.
Image credit: Manos Mavrakis
Carla Silva Martins, François Iv, et al.
Genetically encoded reporters of actin filament organization in living cells and tissues.
Cell 188, 2540-2559 (2025).
https://www.cell.com/cell/abstract/S0092-8674(25)00276-4
Honorable Mention - Video
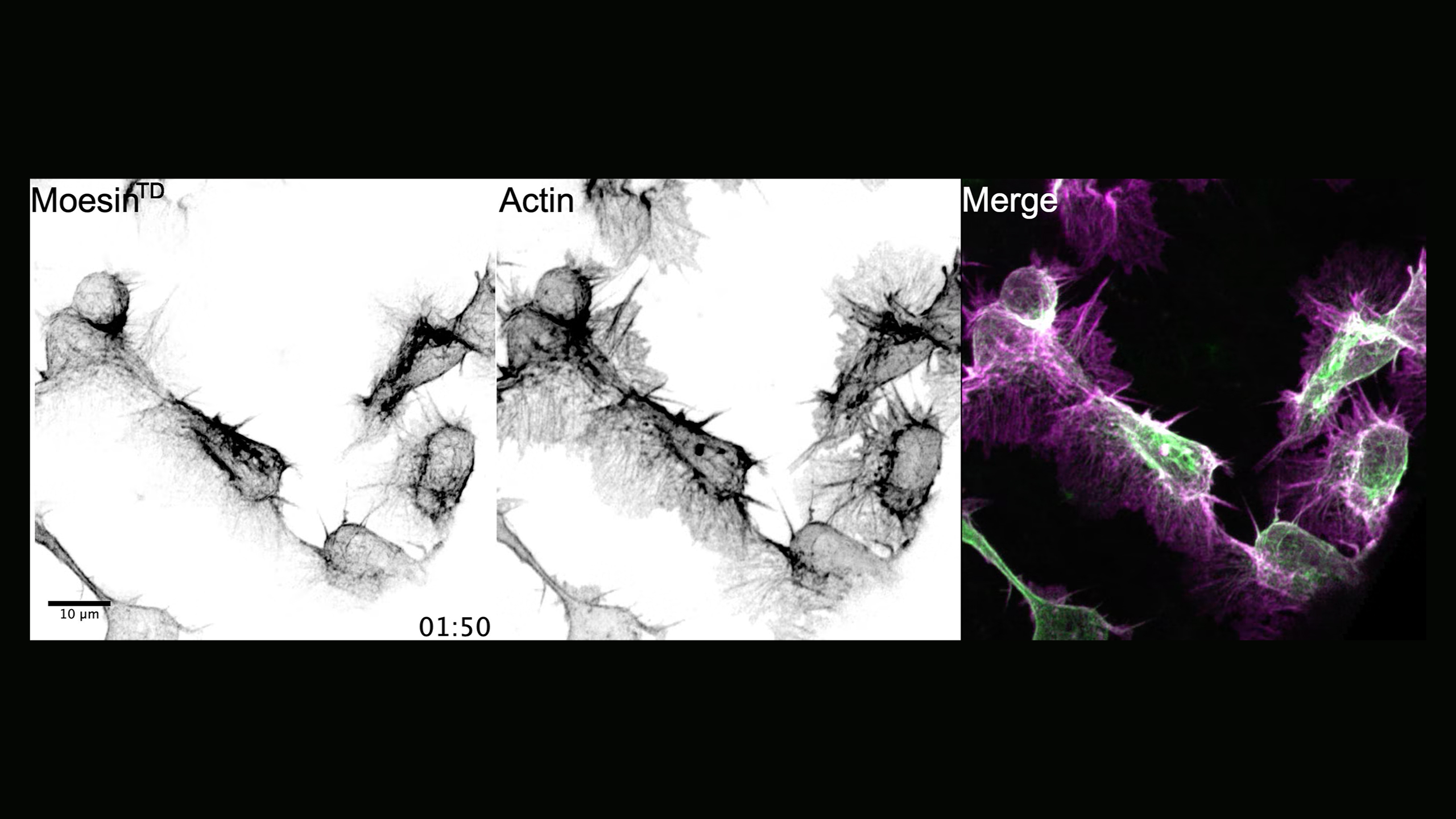
High-resolution imaging of macrophage migration
Time-lapse imaging of macrophages during their embryonic developmental dispersal expressing a single copy of a GFP-tagged phosphomimetic Moesin mutant (MoesinTD), which is constitutively active, while also labelling the actin network. Note the flow of MoesinTD within the lamella towards the cell body and the pulsatile dynamics surrounding the cortex. Time stamp mm:ss.
Video credit: Besaiz J. Sánchez-Sánchez
Besaiz J. Sánchez-Sánchez, Stefania Marcotti, David Salvador-Garcia, María-del-Carmen Díaz-de-la-Loza, Mubarik Burki, Andrew J. Davidson, Will Wood, and Brian M. Stramer.
Moesin integrates cortical and lamellar actin networks during Drosophila macrophage migration.
Nature Communications 16, Article number: 1414 (2025).
https://www.nature.com/articles/s41467-024-55510-5#MOESM12
Honorable Mention - Still Image
Spatial transcriptomics in the adult Drosophila brain and body
The expression of mRNA is regulated locally to allow for cells to differentiate in specific tissues. Spatial transcriptomics can help elucidate this fundamental principle by imaging tens to hundreds of mRNA species in the same sample. We applied spatial transcriptomics to adult Drosophila and selected 6 mRNA species specific to the muscle for this image: in green, muscle myosin; in yellow, Sallimus; in blue and cyan, different Actin isoforms (Act88F and Act79B); and in red and magenta, different Troponin isoforms (TpnC4 and TpnC41C). Interestingly, these isoforms are very specific to a given muscle type, as, for example, Act88F mRNAs (blue) are only located in the flight muscle. Careful attention to the image also shows that Sallimus mRNAs are enriched at the anterior and posterior regions of the flight muscle, and that TpnC4 and Act88F show as bands in the same muscle.
Image credit: Pierre Mangeol
Jasper Janssens, Pierre Mangeol, Nikolai Hecker, Gabriele Partel, Katina I Spanier, Joy N Ismail, Gert J Hulselmans, Stein Aerts, Frank Schnorrer.
Spatial transcriptomics in the adult Drosophila brain and body
2025, Elife 13:RP92618.
https://doi.org/10.7554/eLife.92618.3
Honorable Mention - Video
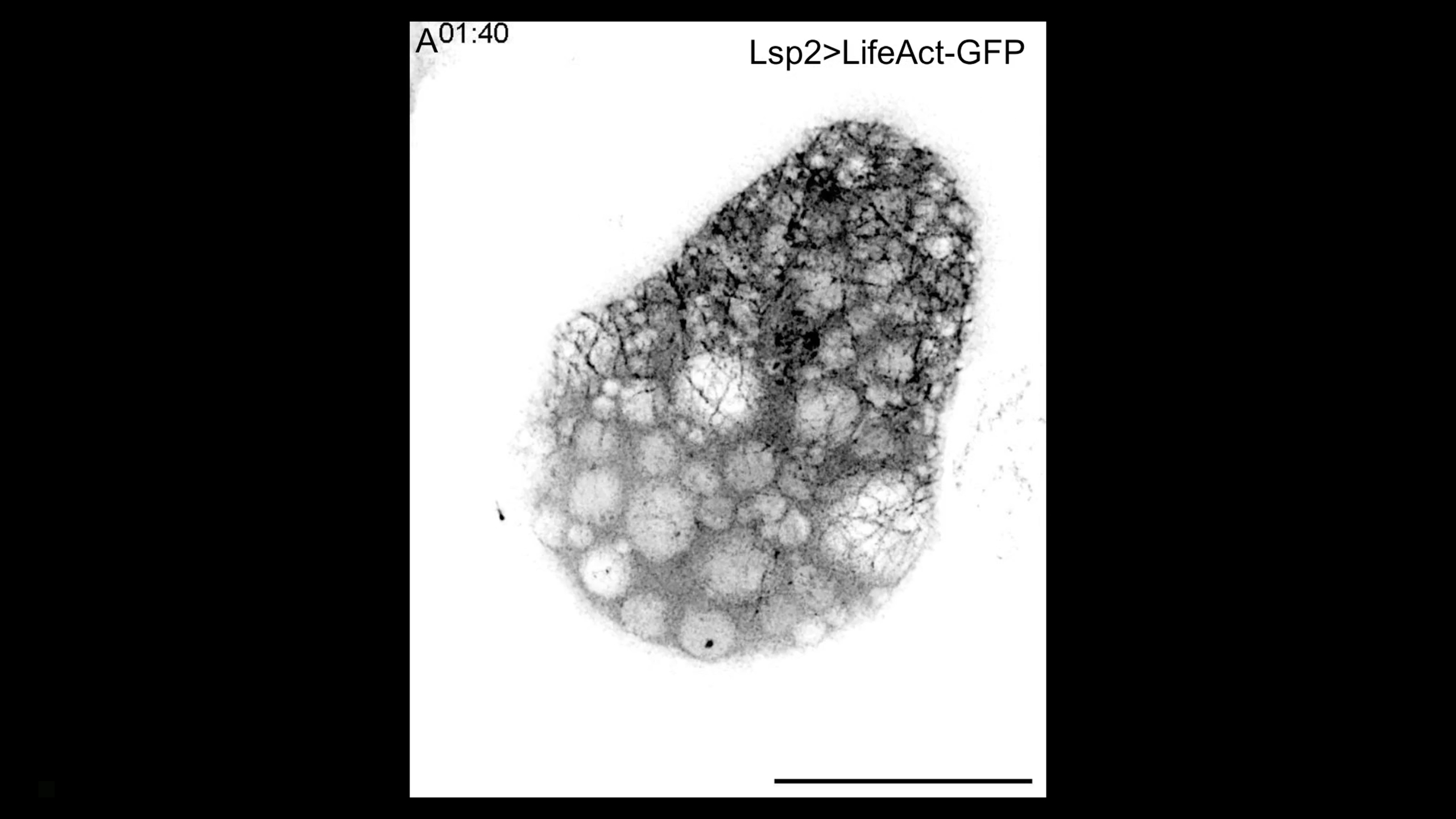
Super-resolution imaging of cortical actin waves in fat body cells during in vivo swimming migration
Complex actin waves drive the in vivo swimming migration of pupal fat body cells. This movie shows two super-resolution microscopy movies visualizing actin waves in swimming pupal fat body cells (Lsp2-Gal4+UAS-LifeAct-GFP). Actin waves propagate along the cell cortex to induce dynamic changes in cell shape. These waves consist of a fibrous actin meshwork composed of highly dynamic actin filaments (red arrowheads) and very long actin bundles connecting actin networks on opposite sides of the cell (blue arrowheads). Elapsed time shown in top left corner in minutes:seconds. Scale bar, 20 μm (full view), 5 μm (zoom).
Video credit: Cyril Andrieu and Anna Franz
Cyril Andrieu, Bren Hunyi Lee and Anna Franz
Cell deformations generated by stochastic actomyosin waves drive in vivo random-walk swimming migration.
J Cell Sci (2025) 138 (10): jcs263787.
https://doi.org/10.1242/jcs.263787
Honorable Mention - Still Image
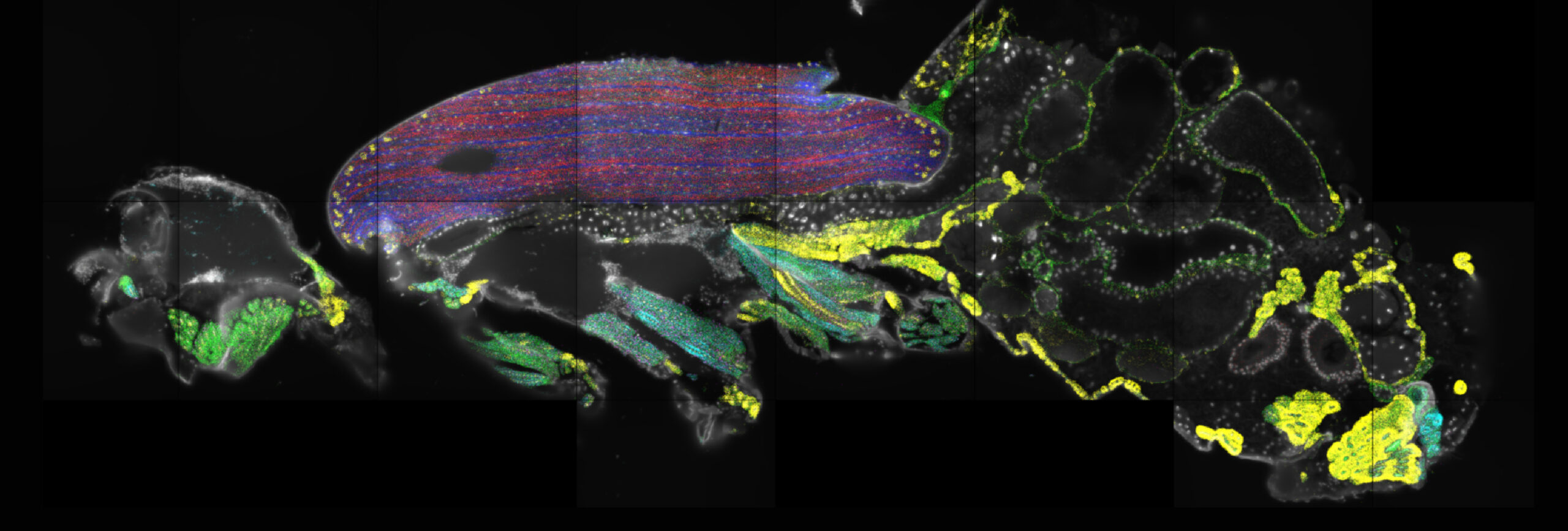
Gut on fire: Tumor metastasis to the fly gut
Tilted confocal image of the adult Drosophila gastrointestinal tract following allografting of a polarity deficient (lgl-/-) neural stem cell–derived tumor line. Metastatic cancer cells (red) disseminate to the midgut, hindgut, and Malpighian tubules and invade the surrounding gut muscle (gray). We show that tumor growth markedly distorts the gut wall but does not breach the epithelial barrier to enter the lumen.
Image credit: Chaitali Khan
Chaitali Khan and Nasser M Rusan.
Allograft transplantation for Drosophila tumor metastasis studies.
Dis Model Mech (2025) 18 (12): dmm052543.
https://doi.org/10.1242/dmm.052543