2005 Drosophila Image Award
Winner
Bubble-like assembly of cells in the developing Drosophila retina.
Surface mechanics constrain cells to adopt physically stable configurations by minimizing the surface free energy. Under this restriction, retinal cone cell clusters self-organize themselves into bubble-like patterns. The figure shows the stable patterns of soap bubble aggregates (left panels) and rough eye (Roi) mutant ommatidia (right panels), where variable numbers of cone cells develop. The extensive correlation suggests that surface minimization is essential for cone cell patterning.
Takashi Hayashi and Richard W. Carthew.
Surface mechanics mediate pattern formation in the developing retina.
Nature 431, 647-652 (2004).
Runner-up
Regulatory evolution at the yellow gene underlies the diversification of wing pigmentation patterns among Drosophilidae.
The adult pigmentation patterns (left) vary from a uniform grey shading such as in Drosophila melanogaster (top) to a unique distal spot (D. biarmipes, middle), to more complex compositions such as in D. guttifera (bottom). The distribution of the protein Yellow during the pupal development revealed here with an antibody against Yellow (right) prefigures these patterns of adult pigmentation. In at least one species, the novel distribution of Yellow is due to the modification of an ancestral enhancer that drives expression in the wing. The evolution of this simple traits provides insights into how animal morphology evolves.
Gompel N.*, Prud’homme B.*, Wittkopp P. J., Kassner V. A. & Carroll S. B. (2005). Chance caught on the wing: cis-regulatory evolution and the origin of pigment patterns in Drosophila.Nature, in press.
* These authors contributed equally to both the paper and the images
Runner-Up
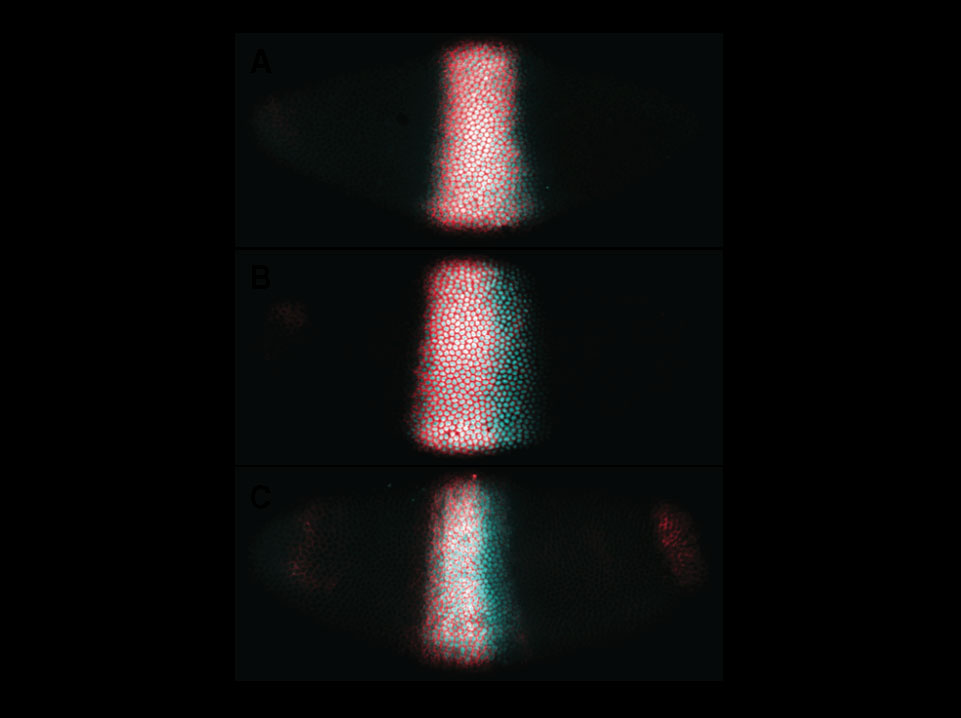
Dynamic shifts of the posterior boundary of the gap gene Krüppelas visualized by asymmetric distribution of RNA and protein expression domains.
Embryos fluorescently stained for both RNA (red) and protein (cyan) products of the gap genes Krüppel are shown at early (A), mid (B) and late (C) cleavage cycle 14A during the blastoderm stage of Drosophila development. In (B) and (C), the RNA domain is located more anteriorly than the protein domain, indicating that Krüppel is only expressed in the anterior portion of the protein domain. Embryo images are aligned such that dorsal is up and anterior to the left.
Jaeger J, Surkova S, Blagov M, Janssens H, Kosman D, Kozlov KN, Manu, Myasnikova E, Vanario-Alonso, CE, Samsonova M, Sharp DH & Reinitz J (2004). Dynamic control of positional information in the early Drosophila embryo. Nature 430: 368-371.
Runner-up
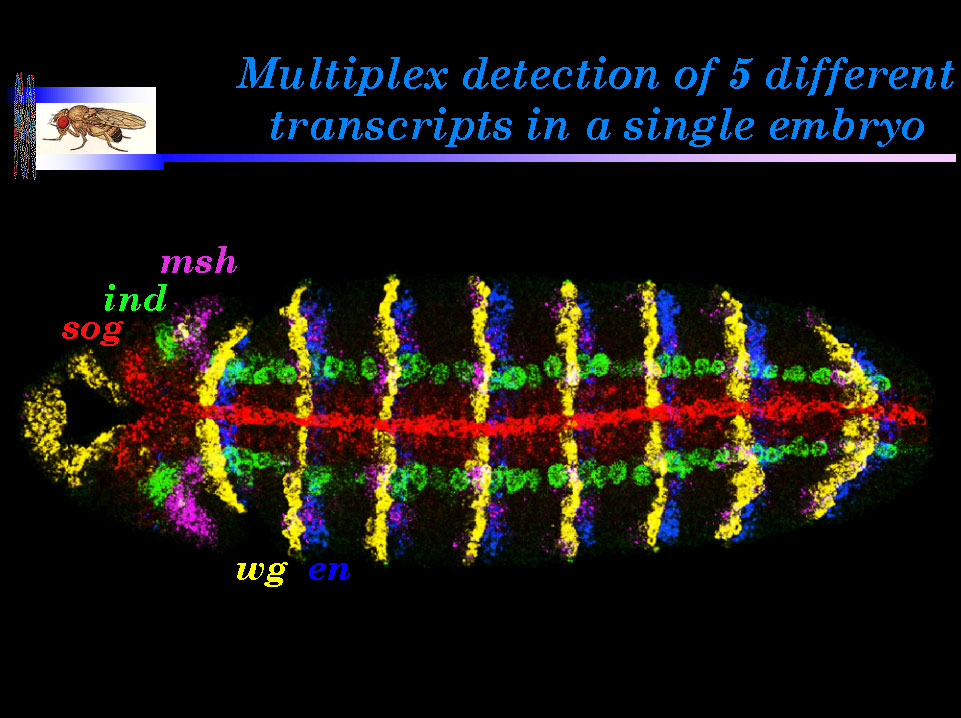
Legend and biological significance
Legend: A ventral view of a germband extended Drosophilaembryo hybridized with five independently labeled probes: sog(direct label, red), ind (green), msh (magenta), wg (yellow) anden (blue).
Biological significance: Multiplex in situ hybridization fills a methodological gap between the existing techniques of genome-wide, but low resolution, RNA profiling, and high resolution single gene in situ detection methods. It is currently possible to detect as many as seven different transcripts in a cell, and by using nascent transcript coding methods, soon it should be possible to detect at least 10-15 transcripts per nucleus (Kosman et al., 2004). This combination of methods should greatly accelerate the construction of gene expression atlases, which will be invaluable in dissecting gene regulatory networks during development and altered patterns of gene expression in disease states.
Kosman, D., C.M. Mizutani, D. Lemons, G. Cox, W. McGinnis, and E. Bier. Multiplex detection of RNA expression in Drosophila embryos. 2004. Science 305, 846.
Finalist
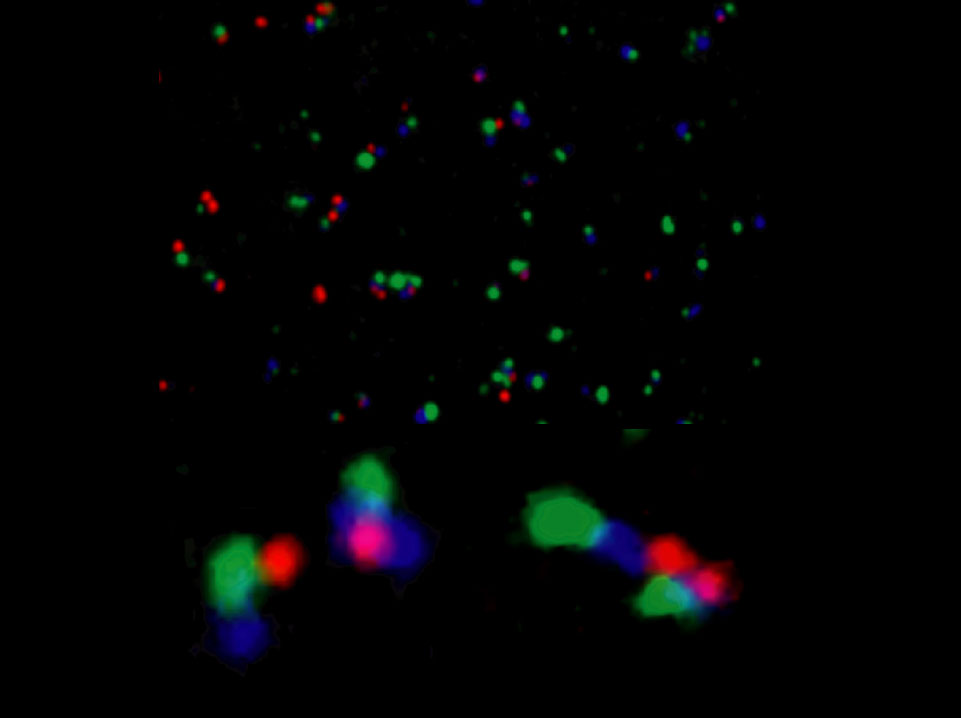
Simultaneous visualization of nascent transcripts from two enhancers and a promoter of the Hox gene Abdominal-B in a Drosophila embryo.
Confocal imaging was used on embryos hybridized with RNA FISH probes for the IAB5 enhancer (green), the IAB8 enhancer (blue), and the Abdominal-B promoter region (red). The upper panel shows a field of nuclei from the A8 region of an elongated embryo hybridized with all three probes. The lower panels shows detailed views of individual nuclei. On the lower left, the signals seen on the chromosome to the left display a nonlinear organization. The promoter region (red) is in close proximity to the distal IAB5 enhancer (green), with the more proximal IAB8 enhancer (blue) displaced away from the promoter. The chromosome on the right displays the expected linear sequence of signals: promoter (BPP, red), proximal IAB8 enhancer (blue), and distal IAB5 enhancer (green). The nascent transcripts associated with IAB8 show a "dumbbell" structure, suggesting that the enhancer is forming a ring near the promoter. The lower right panel details a different nucleus. The chromosome on top shows an extended organization of the Abdominal-B locus, whereas the other homolog (below) is condensed. It appears that the IAB5 enhancer on the compacted chromosome is touching the Abd-B promoter on the extended chromosome.
Matthew Ronshaugen and Mike Levine. Visualization of trans-Homolog Enhancer-Promoter Interactions at the Abd-B Hox Locus in the Drosophila Embryo. 2004. Developmental Cell. 7:925-932
Finalist
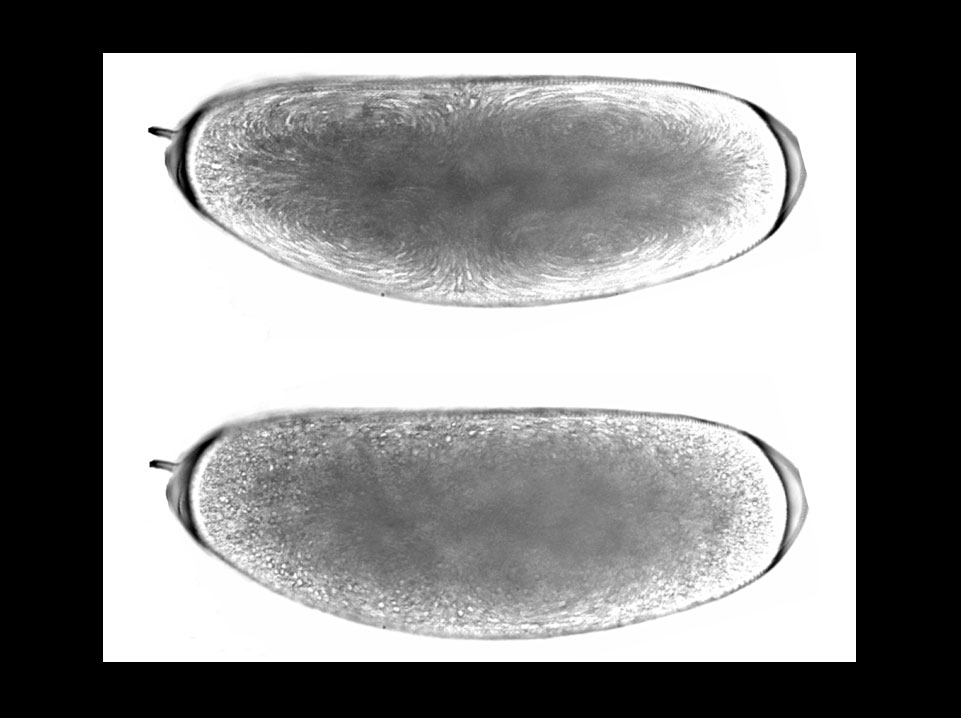
Jun-Yuan Ji* and Gerold Schubiger
Department of Biology, University of Washington, Seattle, WA 98195-1800, USA * Present address: Massachusetts General Hospital Cancer Center, Building 149, 13th Street, Charlestown, MA 02129, USA.
Images are 3-minute projections of a time-lapse recordingduring cycle 6: the upper image shows cytoplamic movements during prophase and metaphase; the lower image shows particle movement during interphase. These 'bright-field transmission' images were generated by allowing the scanning infrared laser beam (900 nm) to pass directly through a live wild-type Drosophila embryo and detecting it using an infrared photodiode.
Ji, J. Y., Squrriell, J. M., and Schubiger, G. (2004). Both Cyclin B levels and DNA-replication checkpoint regulate the early embryonic mitoses in Drosophila. Development 131, 401-411.
Finalist
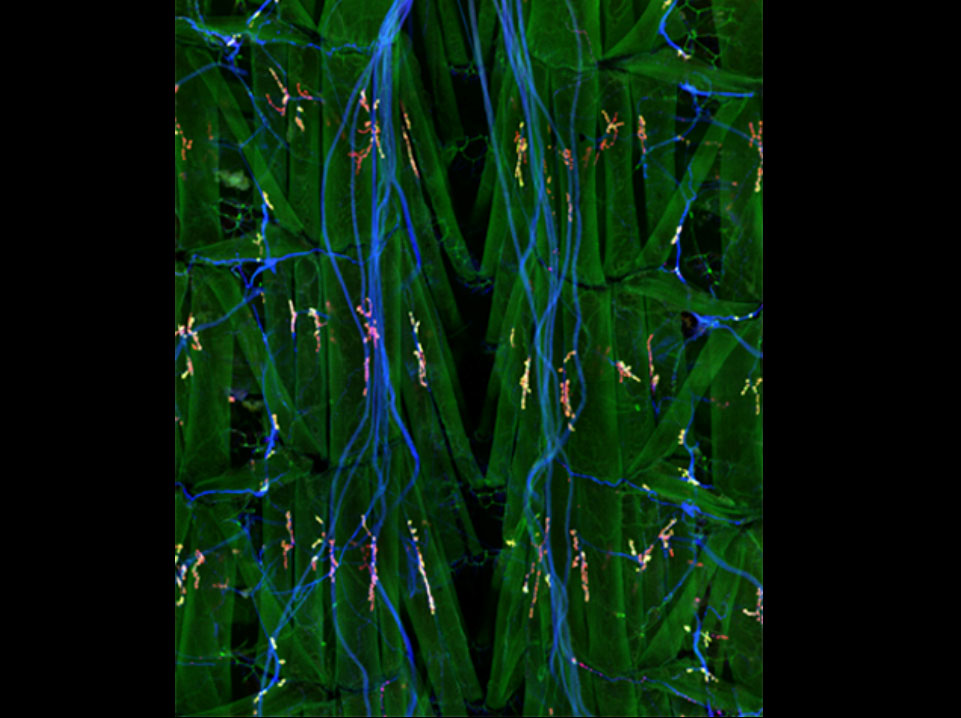
A dissected third-instar Drosophila larvae stained with antibodies to show the synaptic localization of DVGLUT, the Drosophila vesicular glutamate transporter. Neurons are labeled in blue with anti-HRP; muscles and synaptic connections are labeled in green by and antibody to PSD-95 homolog Dlg, and DVGLUT is labeled in red.
Daniels et al., Journal of Neuroscience, 24(46), 10466-10474.
Finalist
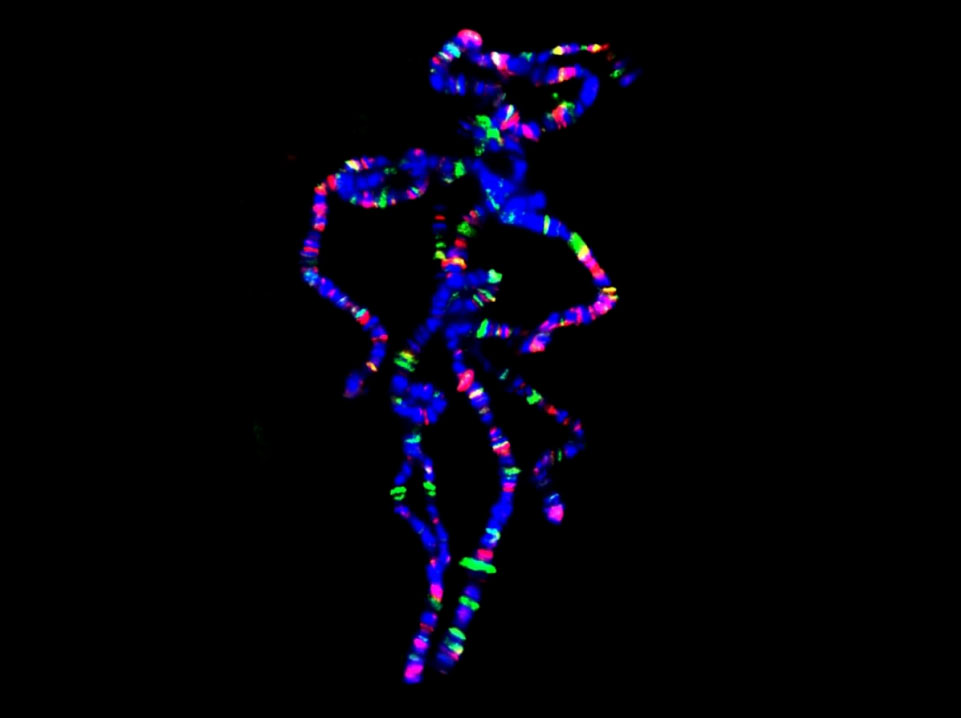
"Pink Panther" chromosomes...
Polytene chromosomes (blue) stained for Hairy (green) and Groucho (red) binding.
(The figure shows the relatively small number of sites binding the transcriptional bHLH repressor Hairy and the surprisingly small overlap (yellow) with its co-repressor Groucho.)
Bianchi-Frias D, Orian A, Delrow JJ, Vazquez J, Rosales-Nieves AE and Parkhurst SM (2004). Hairy Transcriptional Repression Targets and Cofactor Recruitment in Drosophila. PLoS Biology 2, E178.
Finalist
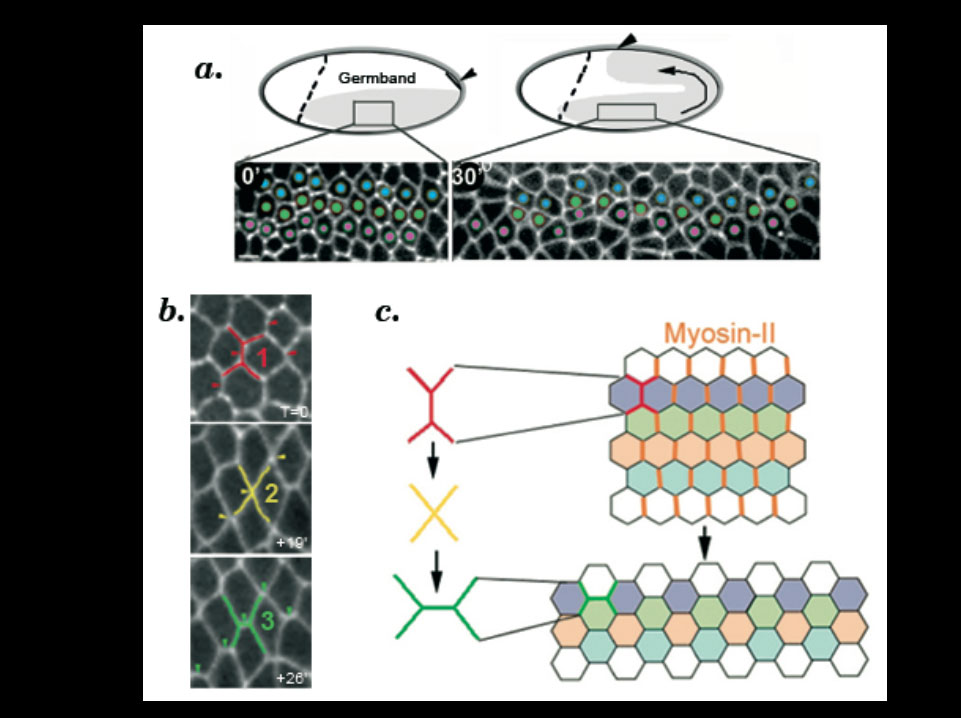
a. Cell intercalation in the ventral-lateral ectoderm (grey) is required for germband elongation. During cell intercalation, cells (marked with a fusion between E-cadherin and GFP) exchange their neighbours.
b. Cell intercalation is a polarized process whereby cell adhesion is remodelled following an irreversible transformation of the geometry of cell contacts. Contacting hexagonal cells progress from a type 1 (red), to type 2 (yellow) and type 3 (green) configuration.c. Polarized junction remodelling is controlled by the specific enrichment of Myosin-II (orange) in type-1 junctions.
Myosin-dependent junction remodelling controls planar cell intercalation and axis elongation.
Bertet. C., Sulak. L., and Lecuit. T. Nature 429:667-671(2004)
Finalist
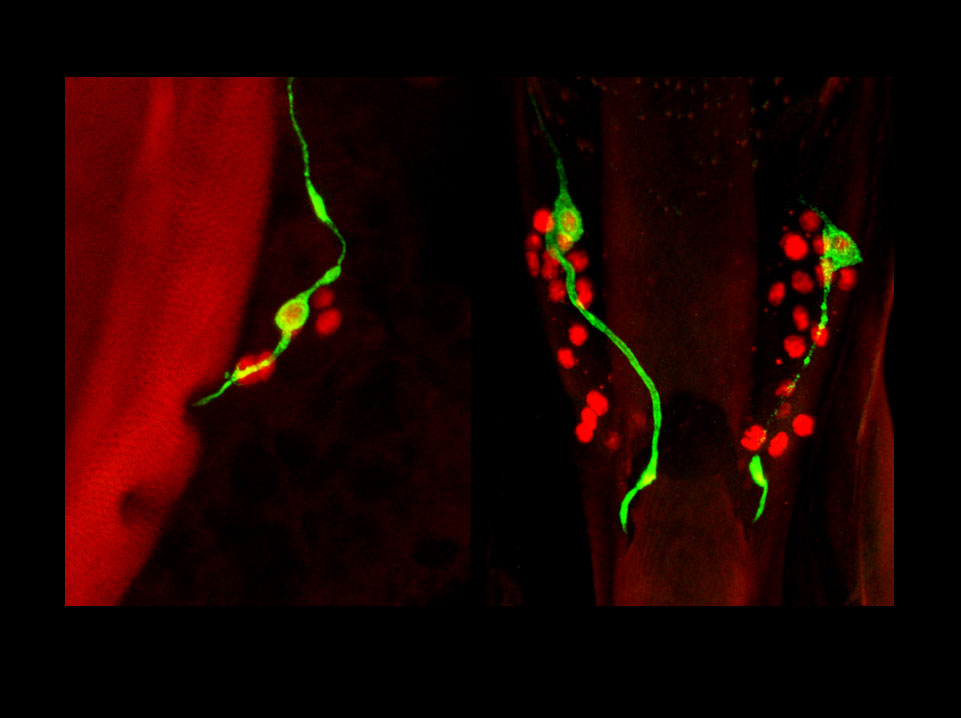
Subsets of larval sensory neurons become integrated in the adult nervous system.
Sense organs of adult Drosophila are essentially formed during metamorphosis. However, evidence presented by Gendre et al. suggests that the gustatory organs on the adult pharynx are mostly persisting larval organs, which were generated during embryonic life. As shown in these confocal images of the pharyngeal sense organs, genetic labeling induced by FLP-out in a neuron-specific P[GAL4] line during late embryogenesis leads invariably to tagging of a single neuron (green) in adult multineuronal sensilla (neuronal nuclei labeled in red). The left and right panels show the dorsal cibarial sense organ and the paired labral sense organs, respectively. Hence, these tagged neurons were born before FLP-out induction. Moreover, their persistence through metamorphosis and integration into the adult nervous system can be directly followed by studying GAL4 expression patterns.
Gendre N, Lüer K, Friche S, Grillenzoni N, Ramaekers A, Technau GM, Stocker RF (2004) Integration of complex larval chemosensory organs into the adult nervous system of Drosophila. Development 131, 83-92.